Advertisements
Advertisements
प्रश्न
Addition of sodium bisulphite to ethanol is ____________ type of reaction.
पर्याय
electrophilic addition
electrophilic substitution
nucleophilic addition
nucleophilic substitution
उत्तर
Addition of sodium bisulphite to ethanol is nucleophilic addition type of reaction.
APPEARS IN
संबंधित प्रश्न
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Phenyl hydrazine
Aldol condensation between which of the following compounds followed by dehydration gives 4-Methylpent-3-en-2-one?
The product formed after reaction between benzaldehyde and methyl magnesium iodide followed by acid hydrolysis is ____________.
Which of the following does not give yellow solid on treatment with sodium hypoiodlte?
Acetone on Clemmensen reduction gives ____________.
Which of the following is a Wolf - Kishner reduction?
Which among the following compounds does NOT undergo aldol condensation?
The formation of cyanohydrin from acetone is an example of ____________.
\[\ce{Benzoic acid ->[i) NH3][ii) \Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\] ‘C’ is:
\[\ce{CH3Br ->[KNC] (A) ->[H3O^+] (B) ->[PCl5] (C)}\] product (C) is:
Identify A, B, C and D.
\[\ce{ethanoic acid ->[SOCl2] A ->[Pd/BaSO4] B ->[NaOH] C ->[][\Delta] D}\]
What is the action of HCN on ethanal?
How is the following conversion effected Hex-3-yne into hexan-3-one?
How is the following conversion effected phenyl methanal into benzoic acid?
How will you prepare ethyl acetate from methyl acetate?
How will you prepare acetophenone from acetyl chloride?
How will you prepare ethane from sodium acetate?
How will you prepare cinnamic acid from benzaldehyde?
Primary nitroalkanes are obtained in good yield by oxidising aldoximes with the help of ______.
Which of the following compound will give positive iodoform test?
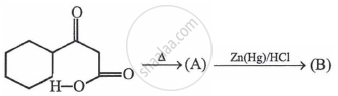
In the above reaction, product (B) is:
Which of the following reaction does not involve either oxidation or reduction?
Aldehydes are readily oxidised to yield carboxylic acids but ketones are inert to oxidation. Which is the most likely explanation regarding this difference in reactivity?
Give two examples which undergo Wolf-Kishner reduction.
Write a note on Clemmensen reduction.
Identify B in the following reaction:
\[\begin{array}{cc}
\ce{O}\phantom{........}\\
||\phantom{........}\\
\ce{\underset{(Acetone)}{CH3 - C - CH3}->[CrO3] B}
\end{array}\]
How will you prepare acetic anhydride from acetic acid?
