Advertisements
Advertisements
प्रश्न
Ammonium salt of benzoic acid is heated strongly with P2O5 and the product so formed is reduced and then treated with NaNO2/HCl at low temperature. The final compound formed is ____________.
पर्याय
Benzene diazonium chloride
Benzyl alcohol
Phenol
Nitrosobenzene
उत्तर
Ammonium salt of benzoic acid is heated strongly with P2O5 and the product so formed is reduced and then treated with NaNO2/HCl at low temperature. The final compound formed is Benzyl alcohol.
APPEARS IN
संबंधित प्रश्न
Write a short note on diazotisation.
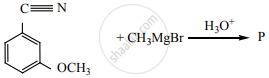
Product ‘P’ in the above reaction is:
The major product of the following reaction:
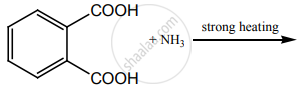
Write a short note on the following.
Coupling reaction
Write a short note on the following.
Gomberg reaction
Account for the following.
Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
Why is \[\ce{NH2}\] group of aniline acetylated before carrying out nitration?
What would be the major product of the following reaction?
\[\ce{C6H5 - CH2 - OC6H5 + HBr -> A + B}\]
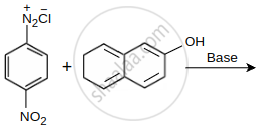
Coupling of benzene diazonium chloride with 1-naphthol in alkaline medium will give:
The major product of the following reaction is: