Advertisements
Advertisements
प्रश्न
An alkyl halide with molecular formula C6H13Br on dehydro halogenation gave two isomeric alkenes X and Y with molecular formula C6H12. On reductive ozonolysis, X and Y gave four compounds CH3COCH3, CH3CHO, CH3CH, CHO and (CH3)2 CHCHO. Find the alkyl halide.
उत्तर
1. C6H13Br is 3 – Bromo – 4 methylpentanc.
\[\begin{array}{cc}\ce{Br}\\
|\\\ce{CH3 - CH - C - CH2 - CH3}\\
|\phantom{......}|\phantom{.......}\\
\ce{CH3}\phantom{...}\ce{H}\phantom{.......}
\end{array}\]
2. 3 – Bromo -4 methylpentane on dehydrogenation give two isomers X and Y as follows:
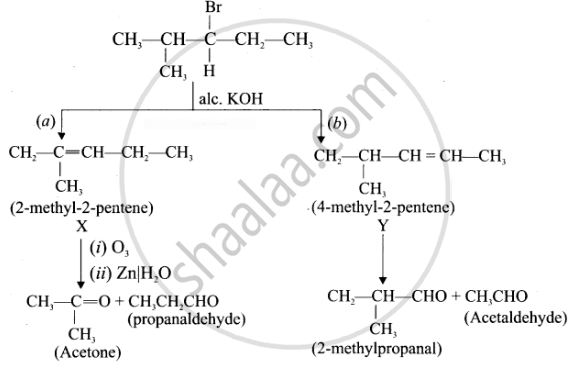
Therefore C6H13 Br is 3 – Bromo – 4 – methy ipentane.
APPEARS IN
संबंधित प्रश्न
Write the reaction to convert propene to isopropyl alcohol.
What is the action of following on but-2-ene?
dil. alkaline KMnO4
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\\phantom{...........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Give IUPAC name for the following compound.
\[\begin{array}{cc}\ce{C2H5}\phantom{.}\ce{CH3}\phantom{.......}\\|\phantom{.....}|\phantom{.........}\\
\ce{CH3 - C - C - C ≡ C - CH3}\\
|\phantom{.....}|\phantom{.........}\\
\ce{CH3}\phantom{..}\ce{H}\phantom{.........}
\end{array}\]
\[\begin{array}{cc}
\phantom{......................................................}\ce{CH3}\phantom{............}\ce{Br}\phantom{}\\
\phantom{....................................................}|\phantom{.............}/\phantom{}\\
\ce{A + Mg ->[Dry][ether] CH3-Mg-Br + B ->[i. Dry ether][ii. H-OH/H^+] CH3-C-OH + Mg}\\
\phantom{.....................................................}|\phantom{.............}\backslash\phantom{}\\
\phantom{......................................................}\ce{CH3}\phantom{..........}\ce{OH}\phantom{}
\end{array}\]
Thus, 'B' in the above reaction is:
Saytzeff's rule states that ____________ product is the major product in dehydrohalogenation of an alkyl halide.
Which of following properties is not exhibited by benzene?
What is the action of following on but-2-ene?
dil alkaline \[\ce{KMnO4}\]
Write the reaction to convert propene to isopropyl alcohol.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH_2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
