Advertisements
Advertisements
प्रश्न
An element X belongs to the 3rd period and 17th group, write the formula of the compound formed when X reacts with \[\ce{_13^27Y}\].
उत्तर
\[\ce{_13^27Y}\] (2, 8, 3)
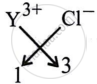
∴ Formula is YBr3.
संबंधित प्रश्न
What is the common name of this group or family?
Write the name and symbol of the element from the description.
The most reactive nonmetal.
Which of the following properties do not match with elements of the halogen family?
Why group 17 elements are highly reactive?
If an element is in Group 17 [or Group 7A] is it likely to be metallic or non- metallic in character?
_______ is in liquid form in the halogen family.
Cl : halogen group : : Ar : _______
Complete the correlation:
Group 1 : Alkali metals : : ______ : Halogens.
Identify the following:
A reaction in which the hydrogen of an alkane is replaced by a halogen.
An element X belongs to 3rd period and 17th group, state the name of the element.
