Advertisements
Advertisements
प्रश्न
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
उत्तर
- In ρ-nitrophenol, nitro group (NO2) is an electron-withdrawing group present at para position which enhances the acidic strength (- I effect). The O – H bond is under strain and release of proton (H+) becomes easy. Further, ρ-nitrophenol ion is more stabilised due to resonance.
- Since the absence of electron-withdrawing group (like –NO2) in phenol at ortho and para position, the acidic strength of phenol is less than that of ρ-nitrophenol.

APPEARS IN
संबंधित प्रश्न
Reaction between Grignard reagent and aldehyde other than formaldehyde leads to formation of _______________
Write the name of major product when anisole reacts HI at 398 K
Draw intramolecular hydrogen bonding structure in o-nitrophenol.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Number of oxygen atoms present in salicylaldehyde are ______.
Which of the following reagents are required in the conversion of phenol to salicylic acid?
Phenol is obtained from cumene ____________.
Sodium metal with ethyl alcohol gives __________ gas.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
The secondary alcohol is ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
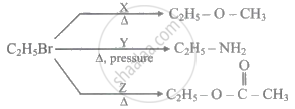
Identify reagents X, Y and Z.
In Raschig's method for synthesis of phenol, the reactants used are ____________.
\[\ce{Isopropyl alcohol + acidic K2Cr2O7 -> X}\]
Identify product 'X' in the above reactions.
Which of the following alcohols is least soluble in water?
Which among the following is allylic secondary alcohol?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify the alcohol that reacts immediately with Lucas reagent?

Product (B) in this reaction is:
Which among the following is not the method of preparation of phenol?
Which of the following compounds reacts immediately with Lucas reagent?
The major product obtained in the following reaction is

Arrange O - H, C - H and N - H bonds in increasing order of their bond polarity.
Explain the reaction of 1° and 2° alcohol with oxidising agent chromic anhydride (CrO3).
Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
