Advertisements
Advertisements
प्रश्न
Answer the following in brief.
Explain graphically the effect of a catalyst on the rate of reaction.
आलेख
टीपा लिहा
उत्तर
i. Diagram:
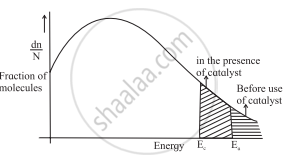
Comparison of fraction of molecules for catalyzed and uncatalyzed reactions
ii. A catalyst lowers the threshold energy.
iii. Consequently, more molecules acquire the minimum amount of energy and tend to cross the energy barrier.
iv. A fraction of activated molecules is greater for the catalyzed reaction. The rate of catalysed reaction thus is larger than the reaction with no catalyst.
shaalaa.com
Effect of a Catalyst on the Rate of Reaction
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
पाठ 6: Chemical Kinetics - Exercises [पृष्ठ १३७]
