Advertisements
Advertisements
प्रश्न
Answer the following in brief.
How will you represent the zeroth-order reaction graphically?
उत्तर
Graphical representation of zero-order reactions: The rate law is given as
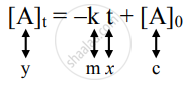
which is a straight line given by y = mx + c.
A plot of `["A"]_"t"` versus t is a straight line.
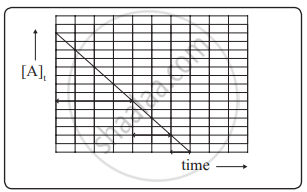
[A]t vs t for zero-order reaction
The slope of the straight line is -k and its intercept on the y-axis is `["A"]_0`.
APPEARS IN
संबंधित प्रश्न
For the reaction 2NOBr → 2NO2 + Br2, the rate law is rate = k[NOBr]2. If the rate of a reaction is 6.5 × 10–6 mol L–1 s–1, when the concentration of NOBr is 2 × 10–3 mol L–1. What would be the rate constant of the reaction?
Explain pseudo first order reaction with a suitable example.
Derive an expression for the relation between half-life and rate constant for first-order reaction.
For a first order reaction \[\ce{A ->Product}\] with initial concentration x mol L−1, has a half life period of 2.5 hours. For the same reaction with initial concentration `("x"/2)` mol L−1 the half life is
Identify the order for the following reaction.
Radioactive disintegration of 92U238
What is the value of rate constant of first order reaction, if it takes 15 minutes for consumption of 20% of reactants?
How long would it take to electroplate a spoon with 0.1 mol of silver (108 g/mol) at a constant current of 2.0 A using AgNO3?
The rate of the reaction \[\ce{A + B -> C}\] is 3.6 × 10−2 mol dm−3 s−1 when [A] = 0.3 mol dm−3 and [B] = 0.2 mol dm−3. Calculate k if reaction is first order in A and zero order in B.
For first order reaction the concentration of reactant decreases from 0.2 to 0.1 M in 100 minutes. What is the rate constant of the reaction?
A first order reaction has rate constant 1 × 10−2 s−1. What time will, it take for 20 g or reactant to reduce to 5 g?
The rate constant of a reaction has same units as the rate of reaction. The reaction is of ____________.
A first order reaction completes its 10% in 20 minutes, then the time required to complete its 19% is ____________.
Which of the following represents the expression for `(3/4)^"th"` life of 1st order reaction?
A first order reaction is 75% completed in 60 minutes, the time required for it's 50% completion is ____________.
For a zero order reaction, the plot of [A]t vs t is linear. The slope of the line is equal to ____________.
Which among the following is an example of zero order reaction?
Which among the following is an example of pseudo first order reaction?
In a reaction \[\ce{N2_{(g)} + 3H2_{(g)} -> 2NH3_{(g)}}\], if the rate of disappearance of N2(g) is 2.6 × 10−4 M/s, the rate of disappearance of H2(g) in M/s is ____________.
For the first order reaction A → B, the rate constant is 0.25 s-1, if the concentration of A is reduced to half, the value of rate constant will be ________.
Half-life of first-order reaction \[\ce{X -> Y + Z}\] is 3 minutes. What is the time required to reduce the concentration of 'X' by 90 % of it's initial concentration?
For zero order reaction, when [A]t is plotted against time (t), the slope of the straight line obtained is equal to ______.
The expression to calculate time required for completion of zero order reaction is ______.
If the rate constant for a first-order reaction is k, the time (t) required for the completion of 99% of the reaction is given by:
Which of the following reactions is not of the first order?
Which among the following reactions is an example of pseudo first order reaction?
The rate constant for a first order reaction is 7.0 × 10-4 s-1. If initial concentration of reactant is 0.080 M, what is the half-life of reaction?
