Advertisements
Advertisements
प्रश्न
Answer the following question.
Why are methane compounds have low melting and boiling points?
उत्तर
Covalent compounds have low intermolecular forces of attraction between the molecules and thus show low melting and boiling points. Since methane is also a covalent compound thus methane has very low melting and low boiling point.
APPEARS IN
संबंधित प्रश्न
Name the scientist who disproved the 'vital force theory' for the formation of organic compounds.
Name the element whose allotropic form is graphite.
Choose the correct alternative
Lamp black is
What is a fire extinguisher ? What is the substance used in the modern type of fire extinguishers ? How is it an improvement over the soda acid-type and the foam-type fire extinguishers ?
FILL IN THE BLANK
.................... is the main gaseous compound of carbon.
Name of the following compounds :
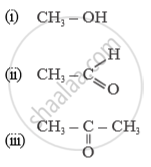
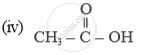
How would you prove experimentally that is slightly acidic in nature.
The property of self-linkage among identical atoms to form long chain compounds is known as ______.
The ability of carbon to form ______ is the main reason for the formation of various classes of carbon compounds.
Give the structure of alcohol with 4 carbon atoms.
