Advertisements
Advertisements
प्रश्न
Answer the following question.
Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidizing agent used in the reaction involved.
उत्तर
In the conversion of ethanol to ethanoic acid, a very strong oxidizing agent alkaline potassium permanganate is used which donates oxygen to the ethanol to form ethanoic acid. Since the addition of oxygen to a compound is termed as oxidation, this reaction is considered as an oxidation reaction.
In this process, the oxidizing agent used is alkaline potassium permanganate.
APPEARS IN
संबंधित प्रश्न
Explain the action of phosphorous trichloride with ethanol. Write the balanced chemical equation of the above reaction.
Fill in the blanks from the choices given within brackets:
The compound formed when ethanol reacts with sodium is____ (sodium ethanoate, sodium ethoxide, sodium propanoate).
Organic compounds having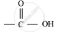 functional group are known as
functional group are known as
Describe one reaction of ethanol.
What is meant by denatured alcohol? What is the need to denature alcohol?
Write a chemical reaction to show the dehydration of ethanol.
Choose the correct word/phrase from the options given below to complete the following sentence:
The product formed when ethene gas reacts with water in the presence of sulphuric acid is ______.
State how the following conversion can be carried out:
Ethyl alcohol to ethene
An organic compound ‘A’ is widely used as a preservative and has the molecular formula C2H4O2. This compound reacts with ethanol to form a sweet-smelling compound ‘B’.
Name the process.
Look at below figure and answer the following questions.

- What change would you observe in the calcium hydroxide solution taken in tube B?
- Write the reaction involved in test tubes A and B respectively.
- If ethanol is given instead of ethanoic acid, would you expect the same change?
- How can a solution of lime water be prepared in the laboratory?
