Advertisements
Advertisements
प्रश्न
Answer the following.
Write a reaction to prepare ethanamine from Propionamide.
उत्तर
Propionamide on Hofmann bromamide degradation yields ethanamine.
\[\begin{array}{cc}\\ \ce{O}\phantom{..........................................................}\\ ||\phantom{..........................................................}\\ \ce{\underset{\text{Propionamide}}{CH3 - CH2 - C - NH2} + Br2 - 4KOH ->[\Delta] \underset{\text{Ethanamine}}{CH3 - CH2 - NH2} + K2CO3 + 2KBr + 2H2O}\\
\phantom{.............}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Answer the following.
Write the IUPAC name of the following amine :
\[\begin{array}{cc}
\ce{CH3 - CH2 - N - CH2 - CH2 - CH3}\\
|\phantom{........}\\
\ce{CH3\phantom{......}}
\end{array}\]
Answer the following.
Write the IUPAC name of the following amine :
\[\begin{array}{cc}\\
\ce{CH3 - CH - NH - CH2 - CH3}\\
|\phantom{...............}\\
\ce{CH3\phantom{............}}
\end{array}\]
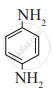
Answer the following.
Write the IUPAC name of the following amine.
H2N-(CH2)6 - NH2
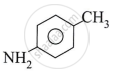
Answer the following.
Write the IUPAC name of the following amine.
Answer the following.
Write a reaction to prepare ethanamine from Nitroethane
The type of isopropylamine is a ______________
Write IUPAC names of trimethylamine
The number of moles of methyl bromide required to convert ethanamine to N, N-dimethylethanamine is ____________.
IUPAC name of the following compound is:
\[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
|\phantom{..}\\
\ce{C2H5 - N - CH3}\phantom{..}
\end{array}\]
IUPAC name of the following compound is:
\[\begin{array}{cc}
\ce{C2H5-N-CH3}\\
|\\
\phantom{...}\ce{CH3}
\end{array}\]
IUPAC name of benzylamine is ______.
IUPAC name of CH3N(C2H5)2 is ________.
Which of the following compounds is obtained on heating primary amine with chloroform and alcoholic potassium hydroxide?
Molecular formula C2H7N represents ____________.
What is the IUPAC name of the following compound?
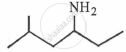
Write the IUPAC name of methylphenylamine.
Write the IUPAC name of benzylamine.
Identify 'B' in the following reactions:
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaCO2/dil.HCl] B.}\]
Write IUPAC name of: