Advertisements
Advertisements
प्रश्न
Answer the questions based on the figure below:
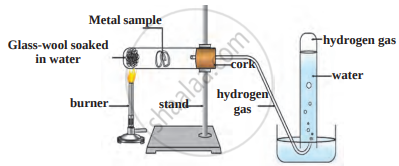
- Which reaction is shown in the figure?
- Which gas is evolved in the reaction?
- Give an example of reactants that rapidly show this reaction. Give equations.
- Give an example of reactants that do not react rapidly.
- In what condition will reactants of (c) part react? Give equation.
उत्तर
- Reaction of a metal with water.
- Hydrogen gas.
- Sodium and potassium metal react rapidly and vigorously with water and liberate hydrogen gas.
i. \[\ce{2Na(s) + 2H2O(l) -> 2NaOH(aq) + H2(g) + Heat}\]
ii. \[\ce{2K(s) + 2H2O(l) -> 2KOH(aq) + H2(g) + Heat}\] - The metals: aluminium, iron and zinc.
- They react with steam to form their oxides. Hydrogen gas is released in this reaction.
i. \[\ce{2Al(s) + 3H2O(g) -> Al2O3(s) + 3H2(g)}\]
ii. \[\ce{2Fe(s) + 4H2O(g) -> Fe3O4(s) + 4H2(g)}\]
iii. \[\ce{Zn(s) + H2O(g) -> ZnO(s) + H2(g)}\]
APPEARS IN
संबंधित प्रश्न
Give two examples of amphoteric oxides.
Name two metals which will displace hydrogen from dilute acids, and two metals which will not.
What are amphoteric oxides?
Give reason:
Sodium, potassium and lithium are stored under oil.
Diamond : electric insulator : : _______ : electric conductor
Example of an amphoteric oxide is:
Which of the following metals liberate hydrogen with 5% HNO3?
(i) Cu
(ii) Zn
(iii) Mn
(iv) Mg
Which one of the following metals does not react with cold as well as hot water?
Metals generally react with dilute acids to produce hydrogen gas. Which one of the following metals does not react with dilute hydrochloric acid?
The metal which produces hydrogen gas on reaction with dilute hydrochloric acid as well as sodium hydroxide solution is
Zinc sulphate forms a colourless solution in water. Will you observe any colour on adding copper turning in it?
Material: Copper wire, Iron nail, Beaker or Big test tube.
Chemicals: Ferrous Sulphate and Copper Sulphate Solution.
- Which reaction you will study with the help of the above material and solutions? Draw the diagram of the experiment arrangement.
- How do you identify that the reaction is carried out?
- Which type of reaction did you observe?
Out of three metals, sodium, calcium and aluminium, which metal reacts most with water? Give the equation of reaction with all three metals.
Answer the questions based on the figure below:
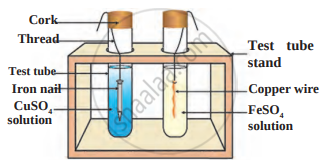
- Which experiment setup is demonstrated in the figure?
- What do you conclude after the reactions? Name reaction.
Select the appropriate state symbols of the products given as X and Y in the following chemical equation by choosing the correct option from table given below:
\[\ce{Zn_{(s)} + H_2SO_{4(l)} ->ZnSO_{4(X)} + H_{2(Y)}}\]
Write a chemical equation showing the ionic products formed on dissolving potassium hydroxide in water.
On adding dilute sulphuric acid to a test tube containing a metal ‘X’, a colourless gas is produced when a burning match stick is brought near it. Which of the following correctly represents the metal ‘X’?
