Advertisements
Advertisements
प्रश्न
- Apurple colour compound A, which is a strong oxidising agent and used for bleaching of wool, cotton, silk and other textile fibres, was added to each of the three test tubes along with \[\ce{H2SO4}\]. It was followed by strong heating.

In which of the above test tubes, A, B or C:
- Violet vapours will be formed.
- The bubbles of gas evolved will extinguish a burning matchstick. Write an equation for each of the above observations.
- A metal ion \[\ce{M^{n+}}\] of the first transition series having d5 configuration combines with three didentate ligands. Assuming Δ0 < P:
- Draw the crystal field energy level diagram for the 3d orbital of this complex.
- What is the hybridisation of \[\ce{M^{n+}}\] in this complex and why?
- Name the type of isomerism exhibited by this complex.
थोडक्यात उत्तर
रासायनिक समीकरणे/रचना
उत्तर
- Test tube C:
\[\ce{10I^- + MnO^-4 + 16 H+ -> 5I2 + 2Mn^{2+} + 8H2O}\] - Test tube A:
\[\ce{C2O^{2-}4 + 2MnO^-4 + 16H+ -> 10CO2 + 2Mn^{2+} + 8H2O}\]
- Test tube C:
-
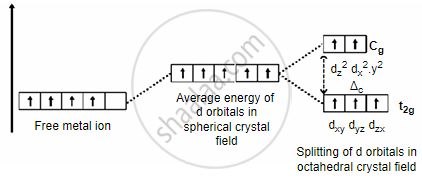
- Sp3d2, Since Δ0 > P, it will form an outer orbital complex as the electrons in the 3d orbital will not pair up.
- Optical isomerism.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
