Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Propan-1-ol, propan-1, 2, 3-triol, propan-1, 3-diol, propan-2-ol
उत्तर
2° alcohols have lower boiling points than 1° alcohol due to a corresponding decrease in the extent of H-bonding because of steric hindrance. Therefore Propan-1-ol has a higher boiling point than Propan-2-ol. Hydrogen group increases, boiling point also increases. Overall increasing order of boiling points is, propan-2-ol < Propan-1-ol < propan-1, 3-diol < propan-1, 2, 3-triol.
APPEARS IN
संबंधित प्रश्न
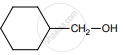 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Among the following ethers which one will produce methyl alcohol on treatment with hot HI?
What is the major product obtained when two moles of ethyl magnesium bromide is treated with methyl benzoate followed by acid hydrolysis.
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Butan-2-ol, Butan-1-ol, 2-methyl propane-2-ol
Is it possible to oxidise t-butyl alcohol using acidified dichromate to form a carbonyl compound?
Explain Kolbe’s reaction.
Complete the following reaction.
\[\ce{C6H5 - CH2CH(OH)CH(CH3)2 ->[conc. H2SO4]}\]
3, 3-dimethyl butane-2-ol on treatment with conc. H2SO4 to give tetramethyl ethylene as a major product. Suggest a suitable mechanism.
\[\begin{array}{cc}\phantom{..........}\ce{CH3}\\\phantom{.......}/\\\ce{C6H4}\\
\phantom{.......}\backslash\\\phantom{.........}\ce{OH}\end{array}\] is a/an ______.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
