Advertisements
Advertisements
प्रश्न
Arrange the following substances in increasing order of forces of attraction between particles -
water, sugar, oxygen
उत्तर
- Sugar is a solid; the forces of attraction between the particles of sugar are strong. Water is a liquid; the forces of attraction here are weaker than sugar. Oxygen is a gas; the forces of attraction are the weakest in gases.
- Thus, the increasing order of forces of attraction between the particles of water, sugar and oxygen is
- Oxygen < Water < Sugar.
APPEARS IN
संबंधित प्रश्न
Give reasons:
We can easily move our hand in air, but to do the same through a solid block of wood, we need a karate expert.
Why do gases have neither a fixed shape nor a fixed volume?
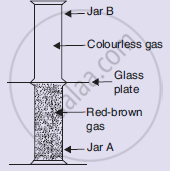
Look at the diagram on the right side. Jar A contains a red-brown gas whereas jar B contains a colourless gas. The two gas jars are separated by a galas plate placed between them
- What will happen when the glass plate between the two jars is pulled away?
- What name is given to the phenomenon which takes place?
- Name the brown gas which could be in jar A.
- Which is the colourless gas most likely to be present in jar B?
- Name one coloured solid and one colourless liquid which can show the same phenomenon.
The substance X normally exists in a physical state which can flow easily but does not fill its vessel completely. It also turns anhydrous copper sulphate blue. When substance X is cooled excessively, it changes into a substance Y which has a fixed shape as well as a fixed volume. If, however, the substance X is heated strongly, it changes into a substance Z which has neither a fixed shape nor a fixed volume.
- Name the substances (i) X (ii) Y and (iii) Z.
- What is the process of conversion of X into Y known as ?
- At which temperature X gets converted into Y ?
- What is the process of conversion of X into Z known as ?
- At which temperature X gets converted into Z ?
Why do gases not have fixed shape?
Comment upon the following:-
kinetic energy
Comment upon the following:-
density
Explain why?
- A gas fills a vessel completely?
- Camphor disappears without leaving any residue.
 |
|
| Fig. I | Fig. II |
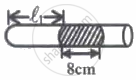
In figure-I, an air column of length ℓ1 is entrapped by a column of Hg of length 8 cm. In figure-II length of same air column at the same temperature is ℓ2. The `ℓ_1/ℓ_2` is:
(1 atm = 76 cm of Hg)
A gaseous mixture of He and O2 is found to have a density of 0.518 gL-1 at 25° C and 720 torr. The mass percent of helium in this mixture is ______.