Advertisements
Advertisements
प्रश्न
- Assertion: Gas is highly compressible than solid and liquid
- Reason: Interatomic or intermolecular distance in the gas is comparably high.
पर्याय
Both the assertion and the reason are true and the reason is the correct explanation of the assertion.
Both the assertion and the reason are true but the reason is not the correct explanation of the assertion.
Assertion is true but the reason is false.
Assertion is false but the reason is true.
उत्तर
Both the assertion and the reason are true and the reason is the correct explanation of the assertion.
APPEARS IN
संबंधित प्रश्न
What do you understand by thermal expansion of a substance?
A metal plate is heated. State three factors on which the increase in its area will depend.
A cubical metal solid block is heated. How will its volume change?
100 ml of each of the following liquid is heated from 10°C to 50°C. Which will expand more : (a) water (b) benzene (c) alcohol?
Distinguish between a clinical and a laboratory thermometer.
Define the following term:
Coefficient of cubical expansion
The diagram shows a sketch graph showing a change in volume of a given mass of ice at -5°C to water at 15°C.
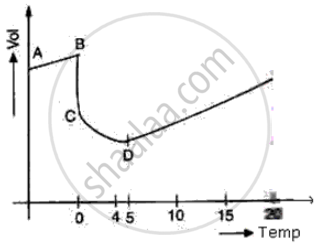
(i) Why does the temperature along BC remain constant?
(ii) When is the volume of water minimum?
(iii) At what temperature does water have maximum density? Explain.
Complete the following sentence:
When ice melts its volume ______ and density _______.
Explain thermal expansion with suitable examples.
