Advertisements
Advertisements
प्रश्न
Assertion: Phenol is more acidic than ethanol.
Reason: Phenoxide ion is resonance stabilized.
पर्याय
both assertion and reason are true and reason is the correct explanation of assertion.
both assertion and reason are true but reason is not the correct explanation of assertion.
assertion is true but reason is false.
both assertion and reason are false.
उत्तर
both assertion and reason are true and reason is the correct explanation of assertion.
APPEARS IN
संबंधित प्रश्न
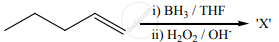
The X is
\[\ce{(CH3)3 - C - CH(OH) CH3 ->[con H2SO4] X (major product)}\]
Write the mechanism of acid catalysed dehydration of ethanol to give ethene.
How is the conversion effected benzyl chloride to benzyl alcohol?
How is the conversion effected benzyl alcohol to benzoic acid?
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following method.
Acid catalysed hydration
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7]A}\]
Predict the major product, when 2-methyl but – 2 – ene is converted into an alcohol in each of the following method.
Acid catalysed hydration.
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Predict the major product, when 2-methyl but-2-ene is converted into an alcohol of the following method:
Acid catalysed hydration
