Advertisements
Advertisements
प्रश्न
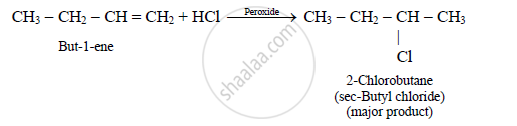
But-1-ene on reaction with HCl in the presence of sodium peroxide yields _______.
पर्याय
n-butyl chloride
isobutyl chloride
secondary butyl chloride
tertiary butyl chloride
उत्तर
secondary butyl chloride
HCl always add according to Markownikoff’s rule even in presence of peroxide
APPEARS IN
संबंधित प्रश्न
Write structures of different dihalogen derivatives of propane.
Give the IUPAC name of the following compound:
CH3C(p-ClC6H4)2CH(Br)CH3
Give the IUPAC name of the following compound:
(CH3)3CCH=CClC6H4I-p
Write the structure of the following organic halogen compound.
2-(2-Chlorophenyl)-1-iodooctane
Write the structure of the following organic halogen compound.
2-Bromobutane
How is nitromethane prepared from the following?
alkyl halide
How is nitromethane prepared from the following?
α-nitroalkene
Write the structure of 1-Bromo-4-chlorobut-2-ene
Out of  which is an example of a benzylic halide?
which is an example of a benzylic halide?
Write the mechanism (using curved arrow notation) of the following reaction:

Write the product(s) in the following reactions:

Complete and rewrite the balanced chemical equations

Write the structures of the main products in the following reactions :
Using IUPAC norms write the formulae for the following:
Hexaamminecobalt(III) sulphate
Using IUPAC norms write the formulae for the following:
Potassium trioxalatochromate (III)
The IUPAC name for tertiary butyl iodide is ____________.
Give IUPAC names of the following compound:
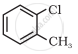
Bromomethane, Chloromethane, Dibromomethane, 1-Chloropropane, Isopropyl chloride, 1-Chlorobutane all are:
Among the following which one can have a meso form?
The compound which contains all the four 1°, 2°, 3° and 4° carbon atoms is:
IUPAC name of CH3CH2C(Br) = CH–Cl is ____________.
Benzene hexachloride is ____________.
Ethylene dichloride can be prepared by adding HCl to ____________.
Benzene diazonium chloride reacts with hypophosphorous acid to produce ____________.
Which of the following is most stable?
\[\ce{CH3CH2CH2Cl ->[alc. KOH] B ->[HBr] C ->[Na/ether] D}\]
In the above reaction, the product D is:
\[\begin{array}{cc}
\ce{CH3-CH-CH=CH2 + HBr -> A}\\
|\phantom{.....................}\\
\ce{CH3}\phantom{..................}
\end{array}\]
'A' is:
Write the structure of the following organic halogen compound.
4-tert. Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Give the IUPAC name of the following compound:
ClCH2C ≡ CCH2Br
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH2C(CH3)2CH2I
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following compound:
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
1, 4-Dibromobut-2-ene
Write the structure of the following compound:
4-tert-Butyl-3-iodoheptane
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Give the IUPAC names of the following compound:
\[\ce{CICH2C ≡ CCH2Br}\]
Write the structures of the following organic halogen compound
1,4-Dibromobut-2-ene
Write structure of the following compound:
1,4-Dibromobut-2-ene
Write structure of the following compound:
4-tert. Butyl-3-iodoheptane
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
Write the structure of the following organic halogen compound:
1,4-Dibromobut 2 ene
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Give the IUPAC name of the following compound:
\[\ce{(CCl3)3CCl}\]
