Advertisements
Advertisements
प्रश्न
Complete and rewrite the balanced chemical equation for the following reactions:
a. \[\ce{1-chloro-2, 4-dinitrobenzene ->[aq. alkali][368 K, H+]}\] ?
b. \[\ce{Benzaldehyde ->[50‰ KOH] }\] ?
c. \[\ce{Acetone + phenylhydrazine ->[H+]}\] ?
टीपा लिहा
उत्तर
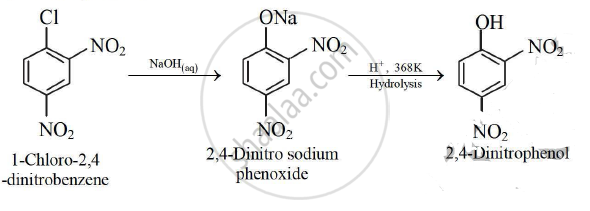
- When l-chloro-2, 4-dinitrobenzene is heated with aqueous alkali (aq. NaOH ), 2, 4-dinitro sodium phenoxide is formed which on hydrolysis with dilute HCl gives 2, 4-dinitrophenol.
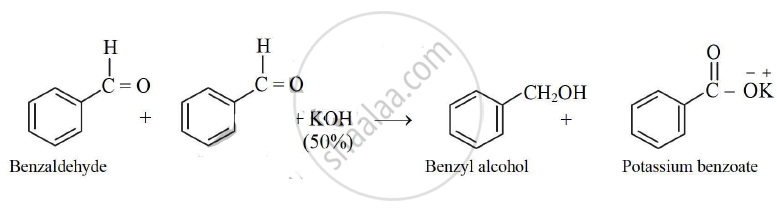
- Two molecules of benzaldehyde react in the presence of 50‰ KOH, the products formed are benzyl alcohol and potassium benzoate. The reaction is called cannizzaro reacion.
- Acetone reacts with phenyl hydrazine to give addition product which on dehydration gives acetone phenyl hydrazone. This reaction occurs in the presence of an acid catalyst.
\[\begin{array}{cc}
\ce{CH3}\phantom{................................}\ce{CH3}\phantom{................................}\\
\backslash\phantom{................................}\backslash\phantom{.............................}\\
\ce{\underset{\text{Phenyl hydrazine}}{C = O + H2N - NH - C6H5} ->[H+] \underset{\text{Acetone phenyl hydrazone}}{C = N - NH - C6H5 + H2O}}\\
/\phantom{................................}/\phantom{............................}\\
\ce{\underset{\text{Aceton}}{CH3}}\phantom{................................}\ce{CH3}\phantom{..............................}
\end{array}\]
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
