Advertisements
Advertisements
प्रश्न
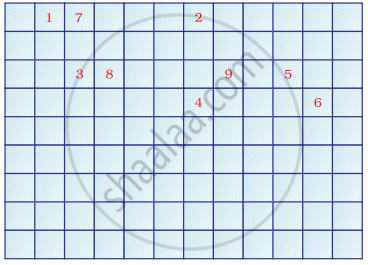
Complete the following cross word puzzle
Across:
(1) An element with atomic number 12.
(3) Metal used in making cans and member of Group 14.
(4) A lustrous non-metal which has 7 electrons in its outermost shell.
Down:
(2) Highly reactive and soft metal which imparts yellow colour when subjected to flame and is kept in kerosene.
(5) The first element of second Period
(6) An element which is used in making fluorescent bulbs and is second member of Group 18 in the Modern Periodic Table
(7) A radioactive element which is the last member of halogen family.
(8) Metal which is an important constituent of steel and forms rust when exposed to moist air.
(9) The first metalloid in Modern Periodic Table whose fibres are used in making bullet-proof vests
उत्तर
Across:
(1) Magnesium
(3) Tin
(4) Iodine
Down:
(2) Sodium
(5) Lithium
(6) Neon
(7) Astatine
(8) Iron
(9) Boron
APPEARS IN
संबंधित प्रश्न
The element which forms a basis oxide has the atomic number of:
(a) 18
(b) 17
(c) 14
(d) 19
The element X forms a compound X2Y. Suggest an element that Y might be and give reasons for your choice.
In which block of the modern periodic table are the nonmetals found?
Classify the following elements into Metals and Nonmetals.
S, Mg, Al, P, N, Na.
The number of periods and groups in the periodic table are ______
What is the atomic number of elements of period 3 and group 17 of the Periodic Table?
The elements A, B, and C belong to groups 1, 14, and 17 respectively of the Periodic Table. Which two elements will form ionic compounds?
The atom of an element has electronic configuration 2, 8, 7. To which of the following elements would it be chemically similar?
In which group are inert elements placed?
Which of the following metals have low melting and boiling point?
