Advertisements
Advertisements
प्रश्न
Consider the graph given in figure. Which of the following options does not show instantaneous rate of reaction at 40th second?
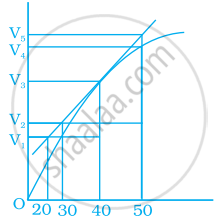
पर्याय
`("V"_5 - "V"_2)/(50 - 30)`
`("V"_4 - "V"_2)/(50 - 30)`
`("V"_3 - "V"_2)/(40 - 30)`
`("V"_3 - "V"_1)/(40 - 20)`
उत्तर
`("V"_4 - "V"_2)/(50 - 30)`
Explanation:
Reaction occurring at the smallest time interval is known as instantaneous rate of reaction. For e.g the instantaneous rate of reaction at 40s is the rate of reaction during a small interval of time close to 40s. Volume changes during a small-time interval close to the 40s.
Instantaneous rate can be determined graphically by drawing a tangent on the curve
Instantaneous reaction = `"Change in concentration"/"Change in time"`
Since the line travel through the graph but does not link to any of the Y-axis lines.
