Advertisements
Advertisements
प्रश्न
Define one kilo-calorie of heat.
उत्तर
One kilo-calorie of heat is the heat energy required to raise the temperature of 1 kg of water from 14.5°C to 15.5°C.
APPEARS IN
संबंधित प्रश्न
Name the mode of transfer of heat in the following :
(a) solid,
(b) liquid,
(c) gas
(d) vacuum
Define the term calorie. How is it related to joule (the S.I. unit of heat)?
1 calorie is 4.81 joules.
Define one calorie.
Ice is kept in a double-walled container. Why?
A circular metal loop is heated at point O as shown in Figure 4.6.
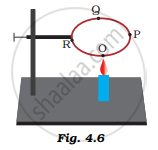
(i) In which direction would heat flow in the loop?
(ii) In which order are the pins at points P, Q and R fixed with the help of wax fall if points O, P, Q and R are equidistant from each other?
Observe the picture given in Figure 4.8. Water is being boiled in a pan of wide base.
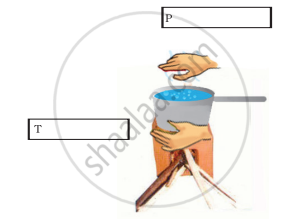
- Which position P or T will feel warmer?
- Fill up the boxes P and T to indicate the mode of flow of heat to the hand.
Which of the following is true?
On a cold day, it is hard to open the lid of a tight container. But when you gently heat the neck you can easily open the lid. why?
Match the following:
| Column A | Column B | ||
| 1. | Heat | a. | Heat gained or lost in the change of state without any change in temperature |
| 2. | m × L. | b. | Heat gained or lost when there is no change of state |
| 3. | Temperature | c. | Form of energy |
| 4. | m × s × t | d. | SI unit of specific latent heat |
| 5. | J/Kg | e. | degree of hotness or coldness |
