Advertisements
Advertisements
प्रश्न
Define the term Aqueous tension
उत्तर
The pressure exerted by saturated water vapour is called aqueous tension.
APPEARS IN
संबंधित प्रश्न
Observe the following conversion.
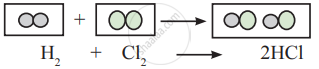
Is the above reaction is in accordance with the principle of stoichiometry?
Observe the following conversion.
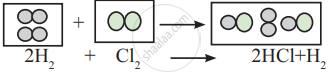
Is the above reaction is in accordance with the principle of stoichiometry?
Define the term polarizability
Define the term Hydrogen bond
Define the term Dipole moment
Identify type of the intermolecular forces in the following compound.
CH3-OH
Identify type of the intermolecular forces in the following compound.
CH2=CH2
Identify type of the intermolecular forces in the following compound.
CH2Cl2
Name the types of intermolecular forces present in Cl2.
Name the types of intermolecular forces present in CCl4.
Name the types of intermolecular forces present in HNO3.
The force of attraction between any two nonpolar molecules is ____________.
Which of the following statements is INCORRECT?
Which of the following is a hydrogen bonded molecular solid?
Which of the following molecules have London forces as predominant intermolecular force of attraction?
The compound incapable of hydrogen bonding with water is ______.
In ____________, hydrogen bonding is stronger than corresponding alcohols.
What type of inter molecular force is present between magnesium chloride and water?
In which of the following compound intra molecular hydrogen bonding is present?
If 'Q' is the magnitude of charge and 'r' is the distance between the centres of positive and negative charges then dipole moment (µ) is given by ______.
Identify the type of intermolecular force present between benzene and ammonia.
