Advertisements
Advertisements
प्रश्न
Describe an experiment to demonstrate that air exerts pressure.
उत्तर
Experiment to demonstrate that air exerts pressure:
Take a thin can fitted with an airtight stopper. The stopper is removed and a small quantity of water is boiled in the can. Gradually the steam occupies the entire space of can by expelling the air from it [Fig (a)]. Then stopper is then tightly replaced and simultaneously the flame beneath the can is removed. Cold water is then poured over the can.
It is observed that the can collapses inwards as shown in fig (b).
The reason is that the pressure due to steam inside the can is same as the air pressure outside the can [Fig (a)]. However, on pouring cold water over the can, fitted with a stopper [fig (b)], the steam inside the can condenses producing water and water vapour at very low pressure. Thus, the air pressure outside the can becomes more than the vapour pressure inside the closed can.
Consequently, the excess atmospheric pressure outside the can causes it to collapse inwards.

APPEARS IN
संबंधित प्रश्न
Explain the following :
A balloon collapses when air is removed from it.
How is water drawn up from a well by a water pump?
A partially inflated balloon is placed inside a bell jar connected to a vacuum pump. On creating vacuum inside the bell jar, balloon gets more inflated. How does the pressure change: increase, decrease or remains same, inside the (a) bell jar, (b) balloon?
How is the reading of a barometer affected when it is taken to: A hill
Define one atmosphere.
Clouds float in atmosphere because of their low ______.
What is meant by atmospheric pressure?
What does low pressure and high pressure lead to?
It is much easier to burst an inflated balloon with a needle than by a finger. Explain.
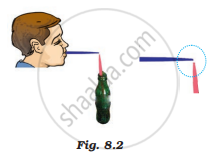
Figure 8.2 shows a child blowing air with a straw near the opening of another straw which has its other end in a soft drink bottle. It was observed that the level of the soft drink in the straw rises up as soon as air is blown over its open end. Which one of the following best explains the reason for rising in the level of the drink?