Advertisements
Advertisements
प्रश्न
Draw a labelled diagram to show electrolytic refining of copper. State what happens when electric current is passed through the electrolyte taken in this case.
थोडक्यात उत्तर
आकृती
उत्तर
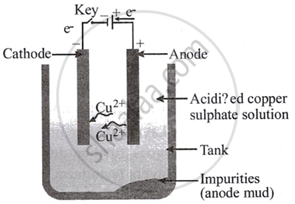
Electrolytic refining of copper
When current flows through the electrolyte, the impure metal dissolves from the anode into the solution. Positive copper ions travel towards the cathode, which is pure copper. Metal cations receive electrons from the cathode and deposit them as \[\ce{Cu}\] atoms, producing pure copper at the cathode.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
