Advertisements
Advertisements
प्रश्न
Elements
उत्तर
To the electronic configuration, a total of 8 electrons must be present in the valence shell of an element. Element
APPEARS IN
संबंधित प्रश्न
Discuss the shape of the following molecules using the VSEPR model:
BeCl2, BCl3, SiCl4, AsF5, H2S, PH3
Although geometries of NH3 and H2O molecules are distorted tetrahedral, bond angle in water is less than that of ammonia. Discuss.
According to VSEPR theory, the repulsion between different parts of electrons obey the order.
Explain VSEPR theory. Applying this theory to predict the shapes of IF7 and SF6.
The H-N-H bond angle in NH3 molecule is ____________.
Consider the species CH4,
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: The H-O-H bond angle in water molecule is 104.5°.
Reason R: The lone pair-lone pair repulsion of electrons is higher than the bond pair-bond pair repulsion.
In the light of the above statements, choose the correct answer from the options given below:
In the following structure, the percentage of the 's' character in the lone pair occupy by the oxygen atom is ______.
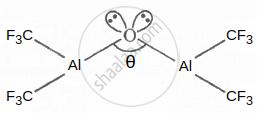
Given: Cos θ = −0.99
What is the number of lone pair of electrons in IF7?
Identify compound having square pyramidal-shape:from following.
