Advertisements
Advertisements
प्रश्न
Explain briefly, how gases can be liquefied.
उत्तर
Gases can be liquefied by applying high pressure and lowering the temperature. Lowering the temperature decreases the kinetic energy of molecules of gas, and increasing the pressure, decreases the space between these particles. Both these effects, cause the molecules to come closer and lead to increase in the intermolecular force of attraction between these molecules. As a result, gas converts into liquid.
APPEARS IN
संबंधित प्रश्न
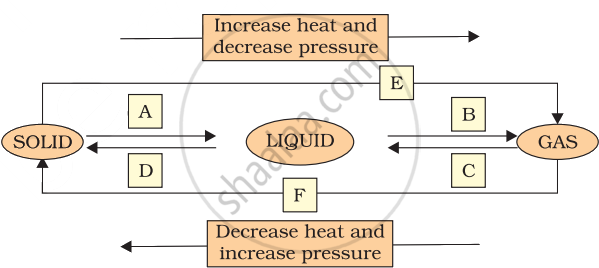
Name A, B, C, D, E and F in the following diagram showing change in its state.
State whether the following is physical or chemical changes.
heating of water —
Draw a labelled diagram of the experimental set-up to study the latent heat of fusion of ice.
Define the term 'latent heat of vaporisation' of a liquid. What is the value of the latent heat of vaporisation of water ?
When water at 0°C freezes to form ice at the same temperature of 0°C, then it :
There are large intermolecular gaps in
A substance which can change its state
Think and try to find a way to demonstrate water cycle in class
Fill in the blanks with the correct words of the statement given below:
During sublimation, a solid on heating turns directly into _________ and on cooling back to __________.
Matter in any state is composed of small particles – molecules, atoms, or ions. Differentiate the terms above in Italics.
