Advertisements
Advertisements
प्रश्न
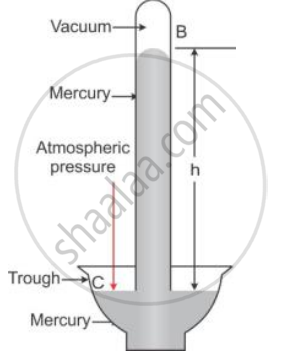
Explain how is the height of mercury column in tube of a simple barometer, a measure of the atmospheric pressure.
उत्तर
In given figure, at all points such as C on the surface of mercury in trough, only the atmospheric pressure acts. When the mercury level in the tube becomes stationary, the pressure inside tube at the point A, which is at the level of point C, must be same as that at the point C. The pressure at point A is due to the weight (or thrust) of the mercury column AB above it. Thus, the vertical height of the mercury column from the mercury surface in trough to the level in tube is a measure of the atmospheric pressure.
The vertical of the mercury column in it (i.e., AB = h) is called the barometric height.
Had the pressure at points A and C be not equal, the level of mercury in the tube would not have been stationary.
APPEARS IN
संबंधित प्रश्न
Give reason for the following:
A balloon collapses when air is removed from it.
What is an aneroid barometer? Draw a neat and labelled diagram to explain its construction and working.
State two factors which affect the atmospheric pressure as we go up.
Give reason:
Air pressure decreases with increasing altitude.
One atmosphere is equivalent to 1,00,000 newton force acting on one square metre.
A low-pressure system is also called ______ and ______.
The highest air pressure at sea level was recorded at ______ Russia.
What does low pressure and high pressure lead to?
Why do Scuba divers wear special suits and equipment?
The increase in internal energy of 1 kg of water at 100°c when it is converted into steam at the same temperature and at 1 atmospheric pressure will be ______.
(density of steam is 0.6 kg m-3, latent heat of vaporisation of water = 2.25 × 106 J kg-1).
