Advertisements
Advertisements
प्रश्न
Explain the relationship between Gibb's standard energy change of the reaction and equilibrium constant.
उत्तर
For the reaction, aA + bB ⇌ cC + dD before approaching equilibrium, the reaction quotient is given by,
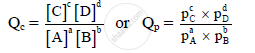
Here, the values of concentrations or partial pressures are other than equilibrium values.
At equilibrium, Q = K called equilibrium constant and ΔG = 0, because the reaction mixture
has no tendency to change in either direction.
Therefore, equation ΔG = ΔG° + RT ln Q becomes
0 = ΔG° + RT ln K
∴ ΔG° = - RT ln K
∴ ΔG° = - 2.303 RT log10K
The above equation gives the relationship between standard Gibbs energy change of the reaction and its equilibrium constant.
APPEARS IN
संबंधित प्रश्न
Amongst the following identify the criterion for a process to be at equilibrium -
- ΔG < 0
- ΔG > 0
- ΔStotal=0
- ΔS < 0
The equilibrium constant Kp for the reaction,
H2(g) + I2(g) → 2HI(g) is 130 at 510 K. Calculate ΔGo for the following reaction at the same temperature: 2HI(g) → H2(g) + I2(g) [Given: R = 8.314 J K-1 mol-1 ]
Derive the relation between `DeltaG^@`and equilibrium constant (K) for the reaction -
aA_bB ⇌ cC+dD.
Write chemical reactions involved in Van Arkel method for refining Titanium
