Advertisements
Advertisements
प्रश्न
Explain the experiment of measuring the real and apparent expansion of a liquid with a neat diagram.
उत्तर
- To start with, the liquid whose real and apparent expansion is to be determined is poured into a container up to a level. Mark this level as L1.
- Now, heat the container and the liquid using a burner as shown in the figure Initially, the container receives the thermal energy and it expands.
- As a result, the volume of the liquid appears to have reduced. Mark this reduced level of liquid as L2.
| Element | No. of Protons | No. of Neutrons | Mass Number | Stable Isotopes (abundance) | Atomic Mass (amu) |
| Nitrogen | 7 | 7 | 14 | N - 14 (99.6%) | 14.004 |
| 7 | 8 | 15 | N - 15 (0.4%) | ||
| Silicon | 14 | 14 | 28 | Si - 28 (92.2%) | 26.04 |
| 14 | 15 | 29 | Si - 29 (4.7%) | ||
| 14 | 16 | 30 | Si - 30 (3.1%) | ||
| Chlorine | 17 | 18 | 35 | CI - 35 (75%) | 35.5 |
| 17 | 20 | 37 | CI - 37 (25%) |
On further heating, the thermal energy supplied to the liquid through the container results in the expansion of the liquid. Hence, the level of liquid rises to L3. Now, the difference between the levels L1 and L3 is called apparent expansion, and the difference between the levels L2 and L3 is called real expansion. The real expansion is always more than that of apparent expansion.
Real expansion = L3 – L2
Apparent expansion = L3 – L1.
APPEARS IN
संबंधित प्रश्न
Explain with the help of formulae the expansion coefficients of liquid and gas.
Explain the following:
Why does not a Pyrex tumbler crack when hot water is poured in it?
A round bottom flask containing coloured water, filtered with a stopper and a narrow glass tube is shown in the following diagram. What will happen to the water level X marked in the tube when it is placed in a hot water bath? Give a reason for your answer.
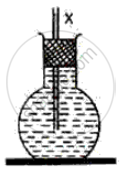
Give reasons for the following:
When boiling water is poured into a glass bottle, it generally cracks.
For a given heat in liquid, the apparent expansion is more than that of real expansion.
