Advertisements
Advertisements
प्रश्न
Explain the geometrical isomerism of the octahedral complex of the type [M(AA)2B2]n± with a suitable example.
थोडक्यात उत्तर
उत्तर
- Consider an octahedral complex of metal M with coordination number six and a bidentate ligand AA and monodentate ligand B having molecular formula [M(AA)2B2]n±
- Bidentate ligand AA has two identical coordinating atoms.
- When two bidentate AA ligands and two "B" ligands are positioned next to each other, a cis-isomer is produced.
- When two B and two AA ligands are positioned opposite each other, a trans-isomer is produced.
- For example, consider a complex [Co(en)2Cl2]+
(a)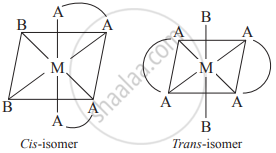
(b)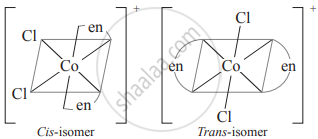
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
