Advertisements
Advertisements
प्रश्न
For the process to occur under adiabatic conditions, the correct condition is ______.
पर्याय
ΔT = 0
Δp = 0
q = 0
w = 0
उत्तर
For the process to occur under adiabatic conditions, the correct condition is q = 0.
Explanation:
A system is said to be under adiabatic conditions if there is no exchange of heat between the system and its surroundings. Hence, under adiabatic conditions, q = 0.
APPEARS IN
संबंधित प्रश्न
Based on the first law of thermodynamics, which of the following is correct?
Consider an isothermal cylinder and massless piston assembly in which ideal gas is filled. Cross-sectional area of the cylinder = 1 m2. Three masses m1, m2 and m3 are kept on the piston. When m1 is removed, the piston moves upto point A. When m1 and m2 both are removed piston moves upto point B and when m1, m2 and m3 all three are removed, piston moves upto point C. The work done by the gas is ______ J, when piston moves from point B to point C.
[Given : m1 = 2 × 104 kg, m2 = 3 × 104 kg, g = 10 m/s2]
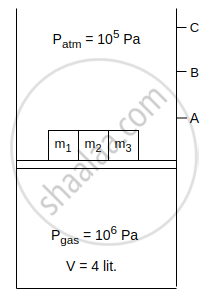
ΔU is equal to ______.
One mole of an ideal gas is expanded isothermally and reversibly to half of its initial pressure. ΔS for the process in JK−1 mol−1 is ______.
[ln 2 = 0.693 and R = 8.314, J/(mol K)]
