Advertisements
Advertisements
प्रश्न
Give a chemical test to distinguish between the following pair of compounds:
Sodium chloride solution and sodium nitrate solution.
उत्तर
To the sodium chloride and sodium nitrate solutions, add the silver nitrate solution. When sodium chloride is present, a curdy white precipitate forms. The reaction mixture stays colourless while dealing with sodium nitrate solution.
APPEARS IN
संबंधित प्रश्न
Potassium sulfite on reacting with hydrochloric acid releases _______ gas. (Cl2, SO2, H2S)
Write an equation for the reaction of aqueous hydrochloric acid on lead nitrate solution.
Give reason for the following:
An aqueous solution of chlorine in acidic in nature
Hint : Cl2 + H2O ⟶ HCl +HClO
Name the following:
Drying agent used to dry hydrogen chloride.
State the use of aqua-regia.
How will you identify?
An amphoteric hydroxide
Identify the salts P and Q from the observation given below:
When dilute HCl is added to a salt Q, a brisk effervescence is produced and the gas turns lime water milky. When NH4OH soltion is added to the above mixture (after adding dilute HCl), it produces a white precipitate which is soluble in excess NH4OH solution.
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Iron (II) sulphide
Write observation:
Lead nitrate solution is mixed with dilute hydrochloric acid and heated.
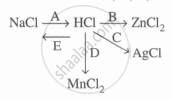
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.