Advertisements
Advertisements
प्रश्न
Give an example of chelate effect.
उत्तर १
\[\ce{Ni^{2+}_{ (aq)} + 6NH_{3 (aq)} <-> \underset{log \beta = 7.99}{[Ni(NH3)6]}^{2+}_{ (aq)}}\]
\[\ce{Ni^{2+}_{ (aq)} + 3en_{(aq)} <-> \underset{(more stable)}{\underset{log \beta = 18.1}{[Ni(en)_3]}}^{2+}_{ (aq)}}\]
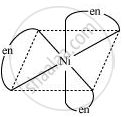
उत्तर २
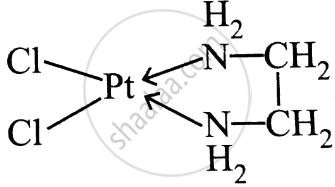
or, \[\ce{[PtCl2(en)]}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
संबंधित प्रश्न
What is meant by the chelate effect?
Answer in brief.
What are the coordination number and oxidation state of the metal ion in the complex [Pt(NH3)Cl5]-?
Answer in brief.
What is the difference between a double salt and a complex? Give an example.
What are the types of ligands? Give one example of each type.
Amongst the following, the ambidentate ligand is ____________
Which among the following is an ambidentate ligand?
What is the number of nitrogen atoms and −COO− groups respectively present in EDTA?
Explain monodentate and ambidentate ligands with example.
Give the steps to understand the metal-ligand bonding.
Write a Example monodentate ligand.
Write the name of a hexadentate ligand.
What are the types of ligands? Give one example of each type.
What are the types of ligands? Give one example of each type.
What are ligands?
What are ligands?
What are the types of ligands? Give one example of each type.
What are ligands?
What are ligands?
