Advertisements
Advertisements
प्रश्न
Give balanced equations for the following reactions:
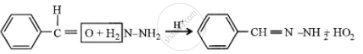
(1) Benzaldehyde reacts with hydrazine.
(2) Acetic acid reacts with phosphorous pentachloride.
(3) Acetone reacts with sodium bisulphite.
उत्तर
(1)
(2) \[\ce{PCI5 + CH3COOH-> CH3COCI + HCI +POCL3}\]
(3) \[\begin{array}{cc}
\ce{CH3\phantom{................} CH3\phantom{..} OH}\\
\phantom{}\backslash \phantom{....................}\backslash \phantom{..}/\\
\ce{C=O + NaHSO3<=>C}\\
\phantom{.}/\phantom{....................}/\phantom{..}|\phantom{...}\\
\phantom{.....}\ce{H3C}\phantom{................}\ce{H3C}\phantom{...}\ce{SO- 3Na+}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
Benzaldehyde undergoes ……….. reaction due to ………… of an α-hydrogen atom.
(Henry’s, aldol condensation, absence, do not, ohm, Raoult’s, increases, common ion effect, easily, three, solubility product, ohm-1, two, four, ohm-1, cm2, Cannizzaro, ohm-1 cm-1, zero, decreases, presence)
How can the following conversions be brought about:
Benzene to benzene diazonium chloride.
Give the balanced equation for the following:
Benzaldehyde with concentrated sodium hydroxide.
