Advertisements
Advertisements
प्रश्न
Give the names and formulae of two strong acids and two weak acids.
उत्तर
The names and formulae of two strong acids and two weak acids are as follows.
(a) Strong acids:
1. Hydrochloric acid: HCl 2. Sulphuric acid: H2SO4
(b) Weak acids:
1. Formic acid: HCOOH
2. Acetic acid: CH3COOH
APPEARS IN
संबंधित प्रश्न
Nitric acid turns red litmus blue.
Name an indicator which is pink in alkaline solution but turns colourless in acidic solution.
When a solution is added to a cloth strip treated with onion extract, then the smell of onion cannot be detected. State whether the given solution contains an acid or a base.\
What happens to temperature of the solution, when an acid is neutralised?
Write the neutralisation reaction between acids and bases in terms of the ions involved.
Give the reaction of acid with nitrates. State the conditions under which they react.
Acid - Define.
What are the uses of acids?
Fill in the crossword given in Figure 5.2 with the help of the clues provided.
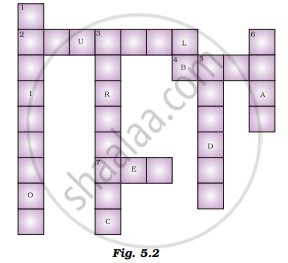
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour to the basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
______ acids have a relatively smaller amount of acids dissolved in a solvent.
