Advertisements
Advertisements
प्रश्न
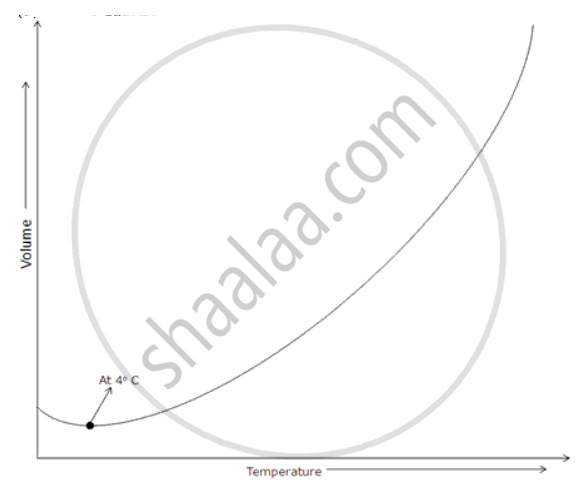
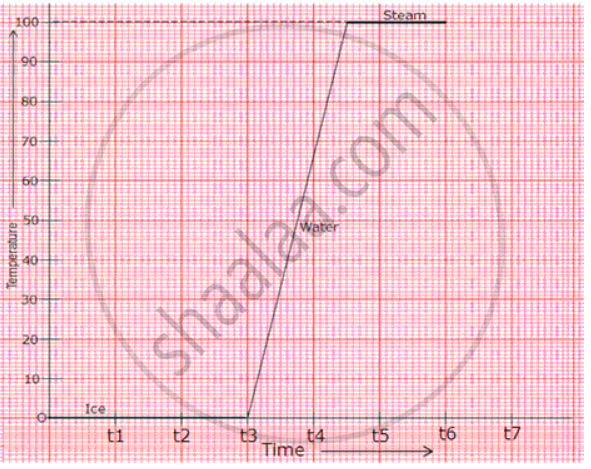
Heat is supplied at a constant rate to 200 g of ice at 0°C until finally all the ice is converted into steam at 100°C. If the specific latent heat of ice is 3.3 x 105 J/kg and that of steam is 2.3 x 106 J/kg and the Sp. heat capacity of water is 4200 J/kg, draw rough graphs for the following to illustrate the changes which take place as the solid ice is converted into water and finally into steam, the pressure remaining constant all the time.
(a) Volume against temperature,
(b) Temperature against time.
आलेख
उत्तर
(a) Volume against temperature
(b) Temperature against time
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
