Advertisements
Advertisements
प्रश्न
How does the anomalous expansion of water help aquatic organisms in cold climates?
उत्तर १
The anomalous expansion of water helps in the survival of water animals in very cold climates. Initially when the temperature of waterfalls, becomes heavier and sinks down. This process continues till 4°C. Then after this expansion takes place. The surface layer of water gets freezed. Ice being a bad conductor of heat does not allow loss of heat from the water below and results in the survival of water animals.
उत्तर २
When water is cooled it contracts and its density, increases until the temperature reaches 4°C, on further cooling water starts expanding with decrease in its density. This is anomalous phenomenon. The aquatic organisms can live there easily.
APPEARS IN
संबंधित प्रश्न
Explain the following:
What is the role of the anomalous behaviour of water in preserving aquatic life in regions of cold climate?
Explain the following:
In cold regions in winter, the rocks crack due to anomalous expansion of water.
A 50 kg man is running at a speed of 18 km h−1. If all the kinetic energy of the man can be used to increase the temperature of water from 20°C to 30°C, how much water can be heated with this energy?
Density of water is maximum at :
Explain, why are soft drink bottles not completely filled?
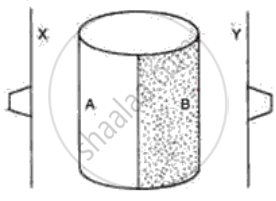
The following figure shows a metal cylinder, containing boiling water. One half side A is polished and another half, B is painted black. Two thin metal sheets X and Y are painted black and have one rubber stopper fixed with wax on each sheet. These sheets are equidistant from the boiling water (container A, B) as shown in the diagram. What would you expect to happen after a few minutes? Give a reason for your answer.
What will be the approximate temperature of water in the lake shown in the following diagram?
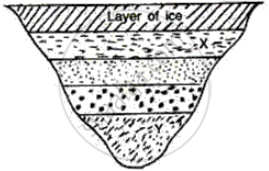
(i) At X, and
(ii) At Y?
Write scientific reason.
Fish can survive even in frozen ponds in cold regions.
