Advertisements
Advertisements
प्रश्न
How will the pH of brine (aq. \[\ce{NaCl}\] solution) be affected when it is electrolysed?
उत्तर
\[\ce{NaCl (aq) -> Na^{+} (aq) + Cl^{-} (aq)}\]
Cathode: \[\ce{H2O(l) + e^{-} -> 1/2 H2(g) + OH^{-} (aq)}\]
Anode: \[\ce{Cl^{-} (aq) -> 1/2 Cl2(g) + e^{-}}\]
Overall reaction: \[\ce{NaCl(aq) + H2O(l) -> NaOH(aq) + H2(g) + Cl2(g)}\]
\[\ce{NaOH}\] is a strong base, they turn the brine solution basic and pH will increase.
APPEARS IN
संबंधित प्रश्न
How many electrons flow through a metllic wire if a current of 0·5 A is passed for 2 hours? (Given : 1 F = 96,500 C mol−1)
If 'I' stands for the distance between the electrodes and 'a' stands for the area of cross-section of the electrode, `"l"/"a"` refers to ____________.
What is the SI unit tor electrochemical equivalent?
Zinc can be coated on iron to produce galvanized iron but the reverse is not possible. It is because ____________.
Define anode
`E_(cell)^Θ` = 1.1V for Daniel cell. Which of the following expressions are correct description of state of equilibrium in this cell?
(i) 1.1 = `K_c`
(ii) `(2.303RT)/(2F) logK_c` = 1.1
(iii) `log K_c = 2.2/0.059`
(iv) `log K_c` = 1.1
Can absolute electrode potential of an electrode be measured?
Cell reaction is spontaneous when
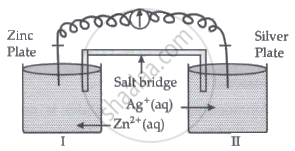
Read the passage given below and answer the questions that follow:
|
Oxidation-reduction reactions are commonly known as redox reactions. They involve transfer of electrons from one species to another. In a spontaneous reaction, energy is released which can be used to do useful work. The reaction is split into two half-reactions. Two different containers are used and a wire is used to drive the electrons from one side to the other and a Voltaic/Galvanic cell is created. It is an electrochemical cell that uses spontaneous redox reactions to generate electricity. A salt bridge also connects to the half-cells. The reading of the voltmeter gives the cell voltage or cell potential or electromotive force. If \[\ce{E^0_{cell}}\] is positive the reaction is spontaneous and if it is negative the reaction is non-spontaneous and is referred to as electrolytic cell. Electrolysis refers to the decomposition of a substance by an electric current. One mole of electric charge when passed through a cell will discharge half a mole of a divalent metal ion such as Cu2+. This was first formulated by Faraday in the form of laws of electrolysis.
|
- Is silver plate the anode or cathode? (1)
- What will happen if the salt bridge is removed? (1)
- When does electrochemical cell behaves like an electrolytic cell? (1)
- (i) What will happen to the concentration of Zn2+ and Ag+ when Ecell = 0. (1)
(ii) Why does conductivity of a solution decreases with dilution? (1)
OR
The molar conductivity of a 1.5 M solution of an electrolyte is found to be 138.9 S cm2mol-1. Calculate the conductivity of this solution. (2)
The cell constant of a conductivity cell is 0.146 cm-1. What is the conductivity of 0.01 M solution of an electrolyte at 298 K, if the resistance of the cell is 1000 ohm?