Advertisements
Advertisements
प्रश्न
How will you carry out the following conversions:
(i) Ethanol to ethene
(ii) Bromoethane to ethanol
उत्तर
(i) Ethanol to ethene:
\[\ce{C2H5OH ->[conc.H2SO4][170°C] CH2 = CH2 + H20}\]
(ii) Bromoethane to ethanol:
\[\ce{C2H5Br + KOH(aq) -> C2H5OH + KBr}\]
APPEARS IN
संबंधित प्रश्न
How can ethanol and ethanoic acid be differentiated on the basis of their physical properties?
Write balanced chemical equations for Preparation of ethanol from Ethyl Chloride
Fill in the blanks from the choices given within brackets:
The compound formed when ethanol reacts with sodium is____ (sodium ethanoate, sodium ethoxide, sodium propanoate).
Organic compounds having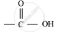 functional group are known as
functional group are known as
What is meant by a functional group? Explain with an example.
What would you see, when ethyne is bubbled through a solution of bromine in carbon tetrachloride?
How will you bring about following conversions:
(a) Methane to methanol
(b) Ethane to ethanol (acetaldehyde)
(c) Methane to methanoic acid
Write a balanced chemical equation for the following:
Ethanol reacts with sodium at room temperature
Convert ethanol into ethene using a solid dehydrating agent. Give only balanced equation.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
