Advertisements
Advertisements
प्रश्न
How will you prepare lactic acid from ethanol?
उत्तर
\[\begin{array}{cc}
\ce{\underset{(Ethanol)}{CH3 - CH2 - OH} ->[(O)] CH3CHO ->[HCN] CH3 - CH - CN ->[H2O][H^+] CH3 - CH - COOH}\\
\phantom{...................................}|\phantom{......................}|\phantom{.}\\
\phantom{.......................................}\ce{OH}\phantom{................}\ce{\underset{(Lactic acid)}{OH}}\phantom{}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write the name of the product when ketones react with 1,2-diol in presence of dry HCl.
ln which of the following reactions, both oxidised and reduced forms of the same compound are obtained?
Which among the following compounds does NOT undergo aldol condensation?
Which one of the following reaction is an example of disproportionation reaction.
Identify X and Y.
\[\ce{CH3COCH2CH2COOC2H5 ->[CH3MgBr] X ->[H3O^+] Y}\]
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How is the following conversion effected Hex-3-yne into hexan-3-one?
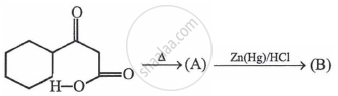
In the above reaction, product (B) is:
Aldehydes are readily oxidised to yield carboxylic acids but ketones are inert to oxidation. Which is the most likely explanation regarding this difference in reactivity?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
