Advertisements
Advertisements
प्रश्न
In an electrophilic substitution reaction of nitrobenzene, the presence of nitro group:
(i) Deactivates the ring by inductive effect.
(ii) Activates the ring by inductive effect.
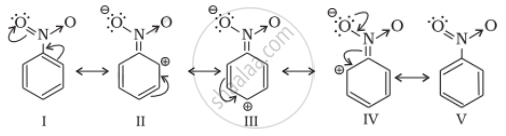
(iii) Decreases the charge density at ortho and para position of the ring relative to meta position by resonance.
(iv) Increases the charge density at meta position relative to the ortho and para positions of the ring by resonance.
उत्तर
(i) Deactivates the ring by inductive effect.
(iii) Decreases the charge density at ortho and para position of the ring relative to meta position by resonance.
Explanation:
The nitro group strongly deactivate the ring due to the -R effect and -I effect
APPEARS IN
संबंधित प्रश्न
Arrange the set of compound in order of their decreasing relative reactivity with an electrophile, E+ Chlorobenzene, 2,4-dinitrochlorobenzene, p-nitrochlorobenzene.
Arrange the set of compound in order of their decreasing relative reactivity with an electrophile, E+ Toluene, p-H3C–C6H4–NO2, p-O2N–C6H4–NO2.
Out of benzene, m–dinitrobenzene and toluene which will undergo nitration most easily and why?
Why do alkenes prefer to undergo electrophilic addition reaction while arenes prefer electrophilic substitution reactions? Explain.
Assertion (A): Nitration of benzene with nitric acid requires the use of concentrated sulphuric acid.
Reason (R): The mixture of concentrated sulphuric acid and concentrated nitric acid produces the electrophile, \[\ce{NO^{+}2}\].
Choose an INCORRECT statement about the electrophilic substitution reaction mechanism from the following:
Excess of isobutane on reactions with Br2 in presence of light at 125°C gives which one of the following, as the major product?
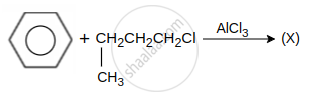
hydrocarbon (X) major product X is:
Benzene on nitration gives nitrobenzene in presence of HNO3 and H2SO4 mixture, where ______.
Which of the following would not give 2-phenylbutane as the major product in a Friedel-Crafts alkylation reaction?
