Advertisements
Advertisements
प्रश्न
In the Cannizzaro reaction given below:
\[\ce{2Ph-CHO ->[OH^-] Ph-CH2OH + PhC\overset{-}{O}_2}\]
the slowest step is:
पर्याय
The attack of OH− at the carbonyl group.
The transfer of hydride to the carbonyl group.
The abstraction of a proton from the carboxylic group.
The deprotonation of Ph–CH2OH.
उत्तर
The transfer of hydride to the carbonyl group.
Explanation:
\[\ce{\underset{Mechanism}{2Ph-CHO} ->[OH^-] Ph-CH2OH + PhC\overset{-}{O}_2}\]
Step I:
\[\begin{array}{cc}
\phantom{...}\ce{O}\phantom{......................}\ce{O^Θ}\phantom{...................}\\
\phantom{}||\phantom{.......................}|\phantom{..................}\\
\ce{Ph - C - H + OH^Θ <=> Ph - C - H}\phantom{...................}\\
\phantom{.......}|\\
\phantom{.........}\ce{\underset{Conjugate base of hydrate of aldehyde}{OH}}
\end{array}\]
Step II:
Intermolecular hydride ioni transfer
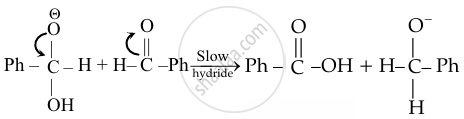
Step III:
\[\begin{array}{cc}
\phantom{}\ce{O}\phantom{..........................}\ce{O}\phantom{..............}\\
\phantom{}||\phantom{..........................}||\phantom{..............}\\
\ce{Ph - C - OH + NaOH -> Ph - C - O^- + Na^+ + H2O}
\end{array}\]
\[\begin{array}{cc}
\phantom{....}\ce{O^-}\phantom{.....................}\ce{OH}\phantom{}\\
\phantom{..}|\phantom{.......................}|\phantom{}\\
\ce{Ph - C - H + H2O -> Ph - C - H}\\
\phantom{..}|\phantom{.......................}|\phantom{}\\
\phantom{..}\ce{H}\phantom{......................}\ce{H}\phantom{}
\end{array}\]
Mechanism H-transfer to the carboxyl group is slow and difficult in this reaction.
