Advertisements
Advertisements
प्रश्न
Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
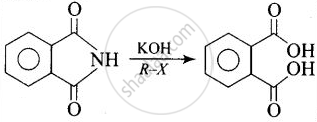
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
उत्तर
| Column I | Column II | ||
| (i) | Ammonolysis | (d) |
Reaction of alkylhalides with \[\ce{NH3}\] \[\ce{R - X -> RNH2 + HCl}\] |
| (ii) | Gabriel phthalimide synthesis | (c) |
Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\]
|
| (iii) | Hoffmann Bromamide reaction | (a) |
Amine with lesser number of carbon atoms \[\ce{RCON2 ->[NaOX] RNH2}\] |
| (iv) | Carbylamine reaction | (b) | Detection test for primary amines. |
APPEARS IN
संबंधित प्रश्न
Give the structures of A, B and C in the following reaction:
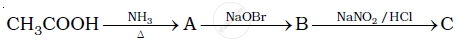
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.

Identify the major product (B).
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following reactions does NOT yield an amine?
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
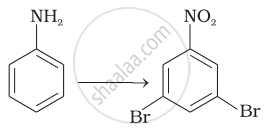
How will you carry out the following conversions?
In the given reaction what is the X?
\[\begin{array}{cc}
\ce{O}\phantom{.......................}\\
||\phantom{.......................}\\
\phantom{}\ce{R - C - OH <-[H3O] Χ ->[H] RCH2NH2}
\end{array}\]
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
Write a short note on the following:
Ammonolysis