Advertisements
Advertisements
प्रश्न
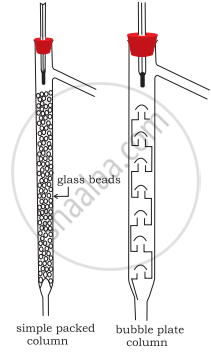
Draw a diagram of bubble plate type fractionating column. When do we require such type of a column for separating two liquids. Explain the principle involved in the separation of components of a mixture of liquids by using fractionating column. What industrial applications does this process have?
उत्तर
Whenever the difference in the boiling points of two liquids is not much than simple distillation technique cannot be used to separate them. The vapours of such liquids are formed within the same temperature range and are condensed simultaneously and hence the technique of fractional distillation is used in such cases. In this technique, vapours of a liquid mixture are passed through a fractionating column before condensation. The fractionating column is fitted over the mouth of the round-bottomed flask. The vapours of the liquid with higher boiling point condense before the vapours of the liquid with lower boiling point. The vapours rising up in the fractionating column become richer in more volatile components and by the time the vapours reach to the top of the fractionating column, these become rich in more volatile components.
APPEARS IN
संबंधित प्रश्न
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more than 25°C), which are miscible with each other?
How would you confirm that a colourless liquid given to you is pure water?
How is distillation more advantageous than evaporation?
Select the correct alternative from the choices given for the following statement:
Components of crude petroleum can be separated by
Explain the difference between Separation by distillation and by separating funnel.
State the correct technique for the separation of the following mixture.
a low boiling point liquid from a high boiling point liquid.
Pure water from impure water
- Give the principle involved in the separation of the mixture
- Give the technique of separation of the mixture.
Choose the correct option.
Which of the following techniques is used for the separation of glycerol from soap in the soap industry?
Alcohol can be separated from water by ______.
Match the type of mixture of compounds in Column I with the technique of separation/purification given in Column II.
| Column I | Column II |
| (i) Two solids which have different solubilities in a solvent and which do not undergo reaction when dissolved in it. | (a) Steam distillation |
| (ii) Liquid that decomposes at its boiling point | (b) Fractional distillation |
| (iii) Steam volatile liquid | (c) Simple distillation |
| (iv) Two liquids which have boiling points close to each other | (d) Distillation under reduced pressure |
| (v) Two liquids with large difference in boiling points. | (e) Crystallisation |
