Advertisements
Advertisements
प्रश्न
Starting with sodium chloride how would you proceed to prepare sodium metal.
उत्तर
Sodium can be extracted from sodium chloride by Downs process.
This process involves the electrolysis of fused NaCl (40%) and CaCl2 (60 %) at a temperature of 1123 K in Downs cell.
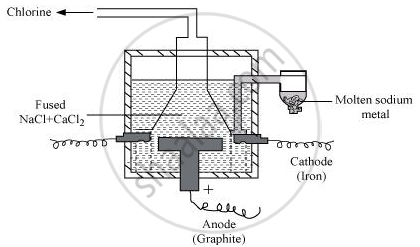
Steel is the cathode and a block of graphite acts as the anode. Metallic Na and Ca are formed at cathode. Molten sodium is taken out of the cell and collected over kerosene.
\[\ce{NaCl ->[Electrolysis] Na+ + Cl-}\]
Molten
At Cathode : \[\ce{Na^+ + e^- -> Na}\]
At Anode : \[\ce{Cl- + e- -> Cl}\]
\[\ce{Cl + Cl -> Cl_2}\]
APPEARS IN
संबंधित प्रश्न
Find the oxidation state of sodium in Na2O2.
Describe two important uses of each of the following caustic soda.
What happens when sodium metal is dropped in water?
What happens when sodium metal is heated in the free supply of air?
State as to why a solution of Na2CO3 is alkaline?
Write a balanced equation for the reaction between Na2O2 and water.
Amphoteric hydroxides react with both alkalies and acids. Which of the following Group 2 metal hydroxides is soluble in sodium hydroxide?
The formula of soda ash is ______.
A chemical A is used for the preparation of washing soda to recover ammonia. When CO2 is bubbled through an aqueous solution of A, the solution turns milky. It is used in whitewashing due to disinfectant nature. What is the chemical formula of A?
Complete the following reactions.
\[\ce{O^{2-}2 + H2O ->}\]
In the Solvay process, can we obtain sodium carbonate directly by treating the solution containing (NH4)2CO3 with sodium chloride? Explain.
Match the elements given in Column I with the colour they impart to the flame given in Column II.
| Column I | Column II |
| (i) Cs | (a) Apple green |
| (ii) Na | (b) Violet |
| (iii) K | (c) Brick red |
| (iv) Ca | (d) Yellow |
| (v) Sr | (e) Crimson red |
| vi) Ba | (f) Blue |
Which of the following metal hydroxides does not dissolve in sodium hydroxide solution?
The pair of compounds which cannot exist together in solution is ______.
