Advertisements
Advertisements
प्रश्न
State and explain Henry’s law
उत्तर
William Henry investigated the relationship between pressure and solubility of a gaseous solute in a particular solvent. According to him, “the partial pressure of the gas in the vapour phase (vapour pressure of the solute) is directly proportional to the mole fraction (x) of the gaseous solute in the solution at low concentrations.” This statement is known as Henry’s law.
Henry’s law can be expressed as,
`"P"_"solute" ∝ "x"_"solute in solution"`
`"P"_"solute" = "KH". "x"_"solute in solution"`
Here, psolute represents the partial pressure of the gas in vapour state which is commonly called as vapour pressure. Xsolute in solution represents the mole fraction of solute in the solution. nature of the gaseous solute and solvent. The above equation is a straight-line in the form of y = mx. The plot partial pressure of the gas against its mole fraction in a solution will give a straight line as shown in fig. The slope of the straight line gives the value of KH.
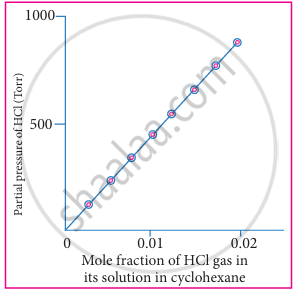 |
| Solubility of HCl gas in cyclohexane at 293 K |
Limitations of Henry’s law:
- Henry’s law is applicable at moderate temperature and pressure only.
- Only the less soluble gases obeys Henry’s law
- The gases reacting with the solvent do not obey Henry’s law. For example, ammonia or HCl reacts with water and hence does not obey this law.
\[\ce{NH3 + H2O <-> NH4+ + OH-}\] - The gases obeying Henry’s law should not associate or dissociate while dissolving in the solvent.
APPEARS IN
संबंधित प्रश्न
Henry’s law constant for the solubility of Nitrogen gas in water at 350 K is 8 × 104 atm. The mole fraction of nitrogen in air is 0.5. The number of moles of Nitrogen from air dissolved in 10 moles of water at 350 K and 4 atm pressure is
Which one of the following gases has the lowest value of Henry’s law constant?
The Henry’s law constants for two gases A and B are x and y respectively. The ratio of mole fractions of A to B 0.2. The ratio of mole fraction of B and A dissolved in water will be
Henry’s law constant for solubility of methane in benzene is 4.2 × 10-5 mm Hg at a particular constant temperature. At this temperature calculate the solubility of methane at
- 750 mm Hg
- 840 mm Hg.
