Advertisements
Advertisements
प्रश्न
Suggest a method to liquefy atmospheric gases.
उत्तर
Atmospheric gases are converted into liquid by increasing the pressure and decreasing the temperature. Method: By filling atmospheric gas in a cylinder, compressing it with a piston attached to it, and reducing the temperature, the particles of the substance come closer and turn into liquid.
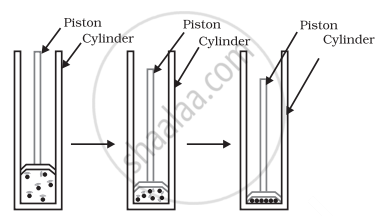
By increasing the pressure the particles of matter can be brought closer.
APPEARS IN
संबंधित प्रश्न
For any substance, why does the temperature remain constant during the change of state?
What is the (a) common unit of temperature, and (b) SI unit of temperature ?
State whether the following statement is true or false :
Solid carbon dioxide is stored under low pressure.
Explain why, ice at 0° C is more effective in cooling than water at the same temperature.
Which contains more heat, 1 kg of water at 100° C or 1 kg of steam at 100° C ? Give reason for your answer.
Define the following term Melting.
Which of the following represent the suitable conditions for the liquefaction of gases ?
On converting 308 K, 329 K and 391 K to Celsius scale, the correct sequence of temperatures will be :
How does a liquid changes into its gaseous state? Explain
Why does this happen?
What you see in the picture.