Advertisements
Advertisements
प्रश्न
The electronic configuration of metal ‘A’ is 2,8,1 and that of metal ‘B’ is 2,8,2. Which of the two metals is more reactive? Write their reaction with dilute hydrochloric acid.
उत्तर
- On moving from left to right in a period of periodic table, the chemical reactivity of elements first decreases from sodium to silicon and then increases from phosphorus to chlorine.
- The electronic configuration of metal ‘A’ is 2, 8, 1. This is electronic configuration of sodium metal.
- The electronic configuration of metal ‘B’ is 2, 8, 2. This is electronic configuration of magnesium metal.
- In the first element of third period, sodium, there is 1 valence electron which it can lose easily to react with other substances, so it is very reactive metal. The second element magnesium has 2 valence electrons. It is not easy for an atom to lose 2 electrons, so magnesium is less reactive than sodium.
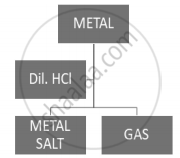
- Reaction with dil hydrochloric acid:
\[2Na + 2HCl \to 2NaCl + H_2 \]
\[Mg + 2HCl \to MgC l_2 + H_2\]
APPEARS IN
संबंधित प्रश्न
Mark ‘T’ if the statement is true and ‘F’ if it is false.
Copper displaces zinc from zinc sulphate solution. ( )
Write the name and formulae of a metal hydride.
Which property of graphite is utilised in making electrodes?
Name two alloys of iron. What elements are present in these alloys?
Give reasons
Aluminium is used in making aircrafts.
State two important uses of following metal
Uses of tin
State two important uses of following metal
Uses of aluminium
State two important uses of following metal
Uses of lead
State the position of the following in the periodic table:
Aluminium
Write name.
The reagent that dissolves noble metals.
Electronic configuration of metal A is 2,8,1. Electronic configuration of metal B is 2,8,8,2. Which of the above metals is more reactive? Explain with reason.
The most abundant metal in the earth’s crust is ____________.
Which of the following two combinations are correct?
| Metal | Gas Evolved | |
| (i) | Copper | Yes |
| (ii) | Iron | Yes |
| (iii) | Magnesium | No |
| (iv) | Zinc | Yes |
The ability of metals to be drawn into thin wire is known as
Give an example of a metal which
- is liquid at room temperature.
- is the best conductor of heat.
Name two major non-metals which are present in fertilisers and enhance the growth of plants.
Why are bells made of metals?
In the given figure, you find that the bulb glows when an iron nail is placed between two ends of wire. Complete the following sentences on the basis of this fact.

- ______ is a metal.
- Metals are good ______ of electricity.
Match the following
| i. | Silica | a. | Caustic potash |
| ii. | Potassium hydroxide | b. | Sand |
| iii. | Sodium hydroxide | c. | Calamine |
| iv. | Zinc carbonate | d. | Caustic soda |
X [2, 8, 7] and Y [2, 8, 2] are two elements. Using this information, complete the following:
Metal atoms tend to have a maximum of ______ electrons in the outermost shell.
