Advertisements
Advertisements
प्रश्न
The graph given below shows heat energy supplied against change in temperature when no energy is lost to the surroundings. The slope of this graph will give:
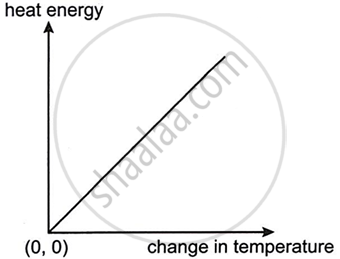
पर्याय
Specific heat capacity
Latent heat of fusion
Latent heat of vaporization
Heat capacity
उत्तर
Heat capacity
Explanation:
Specific heat capacity of a unit mass of substance
= `"Amount of heat energy supplied"/"Difference in temperature"`
APPEARS IN
संबंधित प्रश्न
Why is the base of a cooking pan generally made thick?
What is the principle of the method of mixtures?
Define the term heat capacity.
Name three factors on which heat energy absorbed by a body depends and state how does it depend on them.
The S.I. unit of heat capacity is ______.
Convection is the process by which the thermal energy flows in solids.
Heat capacity C’= ______.
Water is used as a coolant because it ______.
The amount of heat required to raise the temperature to 1°C is called ______.
A solid of mass 60 g at 100°C is placed in 150 g of water at 20°C. The final steady temperature is 25°C. Calculate the heat capacity of solid.
[sp. heat capacity of water = 4.2 J g-1 K-1]
