Advertisements
Advertisements
प्रश्न
The preparation of Lead sulphate from Lead carbonate is a two-step process. (Lead sulphate cannot be prepared by adding dilute Sulphuric acid to Lead carbonate.)
Write the equation for the reaction that will take place when this first step is carried out.
उत्तर
PbCO3 (s) + 2HNO3(dil) → Pb(NO3)2 (aq) + H2O (l) + CO2 ↑
APPEARS IN
संबंधित प्रश्न
Dorji has a few bottles of soft drink in his restaurant. But, unfortunately, these are not labelled. He has to serve the drinks on the demand of customers. One customer wants acidic drink, another wants basic and third one wants neutral drink. How will Dorji decide which drink is to be served to whom?
How will you test for the presence of this gas?
Which chemical is injected into the skin of a person during an ant's sting?
How can the effect of these stings be neutralised?
Define the following term: Acid.
HCl, HNO3, C2H5OH, C6H12O6 all contain H atoms but only HCl and HNO3 show acidic character. Why?
Acids are corrosive in nature.
State whether the following statements are true or false. Correct the false statements.
Lemon water is basic in nature.
Fill in the crossword given in Figure 5.2 with the help of the clues provided.
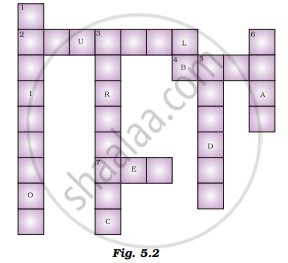
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour to the basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Write the important uses of hydrochloric acid and sulphuric acids.
Complete the following equation.
HCl + H2O → ______ + ______.
